Dispersive solid phase extraction using graphitic carbon nitride microparticles for the determination of trace amounts of lead in water samples
Volume 3, Issue 04, Pages 52-59, Dec 2020 *** Field: Environmental Chemistry
Abstract
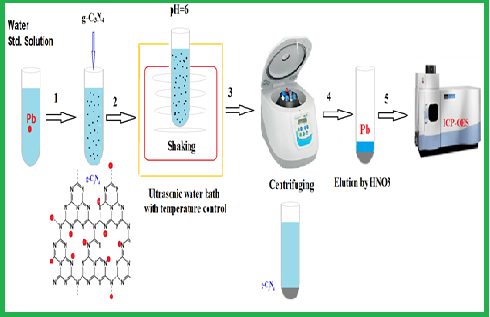
In this work, ultrasound-assisted dispersive micro-solid phase extraction (USA-D-µSPE) technique using graphitized carbon nitride (g-C3N4) is proposed for the preconcentration of low level of lead in aqueous samples. In this method, microparticles of graphitized carbon nitride sorbent were dispersed in the samples using ultrasonic bath and Pb(II) ions were directly adsorbed on the surface of g-C3N4 particles. After adsorption and desorption of lead ions from g-C3N4 particles, the Pb concentration was determined by the inductively coupled plasma- optical emission spectroscopy (ICP-OES). The main advantages of this method are high speed, simplicity and cheapness. The effects of pH, sorbent amount, eluent type and time on the recovery of the analyte were investigated. Under the optimized conditions and preconcentration of 10 mL of sample, the detection limit of 1.24 μg L-1 was obtained. The results were validated by standard reference materials (NIST, SRM) and spiking of real samples by USA-D-µSPE procedure.
References
E.L. Silva, P.d.S. Roldan, Simultaneous flow injection preconcentration of lead and cadmium using cloud point extraction and determination by atomic absorption spectrometry, J. Hazard. Mater., 161 (2009) 142-147.
W.I. Mortada, A.M. Abdelghany, Preconcentration of Lead in Blood and Urine Samples Among Bladder Cancer Patients Using Mesoporous Strontium Titanate Nanoparticles, Biol. Trace Elem. Res., 193 (2020) 100-110.
P.K. Tewari, A.K. Singh, Preconcentration of lead with Amberlite XAD-2 and Amberlite XAD-7 based chelating resins for its determination by flame atomic absorption spectrometry, Talanta, 56 (2002) 735-744.
F. Shah, T.G. Kazi, Naeemullah, H.I. Afridi, M. Soylak, Temperature controlled ionic liquid-dispersive liquid phase microextraction for determination of trace lead level in blood samples prior to analysis by flame atomic absorption spectrometry with multivariate optimization, Microchem. J., 101 (2012) 5–10.
S.R. Yousefi, F. Shemirani, Development of a robust ionic liquid-based dispersive liquid–liquid microextraction against high concentration of salt for preconcentration of trace metals in saline aqueous samples: Application to the determination of Pb and Cd, Anal. Chim. Acta, 669 (2010) 25-31.
K. Saberyan, E. Zolfonoun, M. Shamsipur, M. Salavati-Niasari, Separation and preconcentration of trace gallium and indium by Amberlite XAD-7 resin impregnated with a new hexadentates naphthol-derivative Schiff base, Sep. Sci. Technol., 44 (2009) 1851-1868.
V.O. Vasylechko, G.V. Gryshchouk, M.I. Kaminska, B.M. Stel’makhovych, A solid-phase extraction method using acid-modified Transcarpathian clinoptilolite for preconcentration of trace amounts of lead in water samples, Appl. Nanosci., 9 (2019) 1057-1065.
A. Chisvert, S. Cárdenas, R. Lucen, Dispersive micro-solid phase extraction, TrAC-Trends Anal. Chem., 112 (2019) 226-233.
S.M. Yousefi, F. Shemirani, S.A. Ghorbanian, Deep eutectic solvent magnetic bucky gels in developing dispersive solid phase extraction: Application for ultra trace analysis of organochlorine pesticides by GC-micro ECD using a large-volume injection technique, Talanta 168 (2017) 73-81.
T. Khezeli, A. Daneshfar, Development of dispersive micro-solid phase extraction based on micro and nano sorbents, TrAC-Trends Anal. Chem., 89 (2017) 99-118.
M. Krawczyk, M. Jeszka-Skowron, Multiwalled carbon nanotubes as solid sorbent in dispersive micro solid-phase extraction for the sequential determination of cadmium and lead in water samples, Microchem. J., 126 (2016) 296-301.
A.A. Asgharinezhad, H. Ebrahimzadeh, F. Mirbabaei, N. Mollazadeh, N. Shekari, Dispersive micro-solid-phase extraction of benzodiazepines from biological fluids based on polyaniline/magnetic nanoparticles composite, Anal. Chim. Acta, 844 (2014) 80-89.
J. Wang, Z. Chen, Z. Li, Y. Yang, Magnetic nanoparticles based dispersive micro-solid-phase extraction as a novel technique for the determination of estrogens in pork samples, Food Chem., 204 (2016) 135-140.
E. Zolfonoun, Solid phase extraction and determination of indium using multiwalled carbon nanotubes modified with magnetic nanoparticles, Anal. Method Environ. Chem. J., 1 (2008) 5–10.
F. Aydin, E. Yilmaz, E. Ölmez, M. Soylak, Cu2O-CuO ball like/multiwalled carbon nanotube hybrid for fast and effective ultrasound-assisted solid phase extraction of uranium at ultra-trace level prior to ICP-MS detection, Talanta, 207 (2020) 120295.
E. Zolfonoun, Spectrofluorometric determination of L-tryptophan after preconcentration using multi-walled carbon nanotubes, Anal. Method Environ. Chem. J., 2 (2019) 43-48.
M. Alvand, F. Shemirani, Fabrication of Fe3O4@graphene oxide core-shell nanospheres for ferrofluid-based dispersive solid phase extraction as exemplified for Cd(II) as a model analyte, Microchim. Acta, 183 (2016) 1749-1757.
S.M. Yousefi, F. Shemirani, Carbon nanotube-based magnetic bucky gels in developing dispersive solid-phase extraction: application in rapid speciation analysis of Cr(VI) and Cr(III) in water samples, Int. J. Environ. Anal. Chem., 97 (2017) 1065-1079.
N. Xu, Y. Wang, M. Rong, Z. Ye, Z. Deng, X. Chen, Facile preparation and applications of graphitic carbon nitride coating in solid-phase microextraction, J. Chromatogr. A, 1364 (2014) 53-58.
J. Yang, L. Si, S. Cui, W. Bi, Synthesis of a graphitic carbon nitride nanocomposite with magnetite as a sorbent for solid phase extraction of phenolic acids, Microchim. Acta, 182 (2015) 737-744.
J. Liu, T. Zhang, Z. Wang, G. Dawson, W. Chen, Simple pyrolysis of urea into graphitic carbon nitride with recyclable adsorption and photocatalytic activity, J. Mater. Chem., 21 (2011) 14398-14401.
H.B. Zheng, J. Ding, S.J. Zheng, G.T. Zhu, B.F. Yuan, Y.Q. Feng, Facile synthesis of magnetic carbon nitride nanosheets and its application in magnetic solid phase extraction for polycyclic aromatic hydrocarbons in edible oil samples, Talanta, 148 (2016) 46-53.
Copyright (c) 2020 Analytical Methods in Environmental Chemistry Journal

This work is licensed under a Creative Commons Attribution 4.0 International License.
JOURNAL PUBLISHING AGREEMENT
PLEASE PROVIDE US THE FOLLOWING INFORMATION,
Article entitled:
Corresponding author:
To be published in the journal:
Your Status
I am the sole author of the manuscript
- I am an Iranian government employee.
- I am a European government employee
- I am a Asian government
- None of the above
I am one author signing on behalf of all co-authors of the manuscript
- I am an Iranian government employee.
- I am a European government employee
- I am a Asian government
- None of the above
Please tick the above blanks (as appropriate), review the Journal Publishing Agreement, and then sign and date the document in black ink.
Published Journal Article: the author may share a link to the formal publication through the relevant DOI. Additionally theses and dissertations which contain embedded Published Journal Articles as part of the formal submission may be hosted publicly by the awarding institution with a link to the formal publication through the relevant DOI. Any other sharing of Published Journal Articles is by agreement with the publisher only.
Signed: ______________________________________ Name printed: ___________________________________________
Title and Company (if employer representative): _______________________Date: __________________________________