Thallium extraction in urine and water samples by nanomagnetic 4-Aminothieno[2,3-d] pyrimidine-2-thiol functionalized on graphene oxide
Volume 4, Issue 03, Pages 68-79, Sep 2021 *** Field: Analytical Chemistry
Abstract
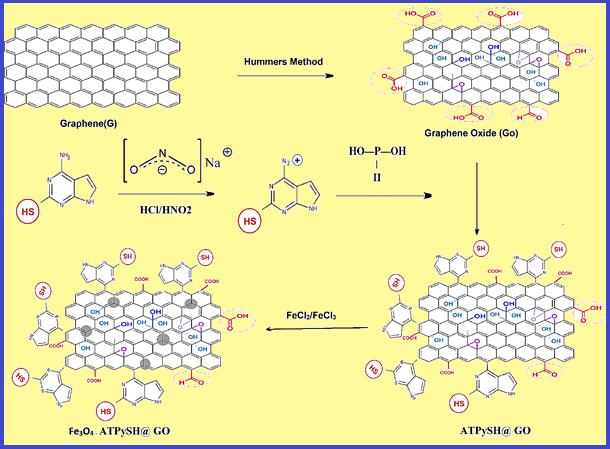
Thallium is a water-soluble metal and extra dosage has toxicological effect in human body. Thallium is readily absorbed by inhalation, ingestion and skin contact. The symptomatology of thallium toxicity was seen in patients with hemorrhage, bone/gastrointestinal problems, delirium, convulsions and coma. So, accurate determination of thallium in water and human urine is necessary. In this research, a novel and applied method based on 25 mg of nanomagnetic 4-Aminothieno[2,3-d] pyrimidine-2-thiol functionalized on graphene oxide (Fe3O4-ATPyHS@GO) was used for thallium extraction in 50 mL of water, wastewater and urine samples by dispersive magnetic micro solid-phase extraction (DM-μ-SPE). After extraction and back-extraction of solid phase by 1 mL of nitric acid solution, the concentration of thallium ions determined by flame atomic absorption spectrometry (F-AAS). The working/linear range, the limit of detection (LOD), and preconcentration factor (PF) were achieved (4-1400 μg L−1; 4-300 μg L−1), 0.9 µg L−1, and 50, respectively (Mean RSD%=1.8 water; 2.1 urine). The absorption capacity of Go and Fe3O4-ATPyHS@GO adsorbent were achieved 7.2 and 137.5 mg g-1 for 5 mg L-1 of thallium, respectively. The procedure was validated by ICP-MS analyzer.
References
T. S. Lin, J.O. Nriagu, Thallium in the environment, New York: Wiley, pp.31-44,1998.
C. Yang, Y. Chen, P. Peng, X. Chang, C. Xie, Distribution of natural and anthropogenic thallium in the soils in an industrial pyrite slag disposing area, Sci. Total Environ., 341 (2005) 159–172.
A. Vaněk, Z. Groesslova, M. Mihaljevic, Thallium contamination of soils/vegetation as affected by sphalerite weathering: a model rhizospheric experiment, J. Hazard. Mater., 283 (2015) 148–156. https://doi.org/10.1016/j. jhazmat.2014.09.018.
M. Sadowska, E. Biaduń, B. Krasnodębska-Ostręga, Stability of Tl(III) in the context of speciation analysis of thallium in plants, Chemosphere, 144 (2016) 1216–1223.
Z. Ning, L. He, T. Xiao, L. Márton, High accumulation and subcellular distribution of thallium in green cabbage, Int. J. Phytoremed., 17 (2015)1097–1104.
B. Krasnodebska-Ostrega, Tl I and Tl III presence in suspended particulate matter: speciation analysis of thallium in wastewater, Environ. Chem., 12 (2015) 374–379.
P. Cvjetko, I. Cvjetko, M. Pavlica, Thallium toxicity in humans, Arch. Ind. Hyg. Toxicol., 61 (2010) 111–119. https://doi.org/10.2478/10004-1254-61-2010-1976.
J.M. Wallace, Kale and thallium: insights from your nutrition team. Permaculture solutions for healing, 2015.
A. Lansdown, The carcinogenicity of metals: human risk through occupational and environmental exposure, Cambridge: Royal Society of Chemistry, pp. 323–330, 2013.
Z. Groesslova, A. Vanek, M. Mihaljevic, V. Ettler, M. Hojdovác, T. Zádorováa, Bioaccumulation of thallium in a neutral soil as affected by solid-phase association, J. Geochem. Explor., 159 (2015) 208–212.
T. Wojtkowiak, B. Karbowska, W. Zembrzuski, M. Siepak, Z. Lukaszewski, Miocene colored waters: a new significant source of thallium in the environment, J. Geochem. Explor., 161 (2016) 42–48. https://doi.org/10.1016/j. gexplo.2015.09.014.
F.S. Hussain, N.S. Hussain, Clinical utility of thallium-201 single photon emission computed tomography and cerebrospinal fluid epstein-barr virus detection using polymerase chain reaction in the diagnosis of AIDS-related primary central nervous system lymphoma, Cureus., 2016 May 8 (2016) e606.
L. Osorio-Rico A. Santamaria S. Galván-Arzate, Thallium toxicity: general issues, neurological symptoms, and neurotoxic mechanisms, Adv. Neurobiol., 18 (2017) 345-353.
V. Yu, M. Juhász, A. Chiang, N. Atanaskova Mesinkovska, Alopecia and associated toxic agents: A systematic review, Skin Appendage Disord., 4 (2018) 245-260.
HY. Yu, C. Chang, F. Li, Q. Wang, M. Chen, J. Zhang, Thallium in flowering cabbage and lettuce: Potential health risks for local residents of the Pearl River Delta, South China, Environ. Pollut., 241 (2018) 626-635.
B. Štádlerová, M. Kolrosová, J. Dědina, S. Musil, Atomic fluorescence spectrometry for ultrasensitive determination of bismuth based on hydride generation – the role of excitation source, interference filter and flame atomizers, J. Anal. At. Spectrom., 35 (2020) 993-1002.
B. Karbowska, T. Rębiś, G. Milczarek, Electrode modified by reduced graphene oxide for monitoring of total thallium in grain products, Int. J. Environ. Res. Public Health, 15 (2018) 653.
L. Nyaba, T. S. Munonde, Magnetic Fe3O4@Mg/Al-layered double hydroxide adsorbent for preconcentration of trace metals in water matrices, Sci. Reports, 11(2021) 2302.
L. Nyaba, B. Dubazana, A. Mpupa, P. N. Nomngongo, Development of ultrasound-assisted dispersive solid-phase microextraction based on mesoporous carbon coated with silica@iron oxide nanocomposite for preconcentration of Te and Tl in natural water systems, Open Chem., 18 (2020) 412–425.
T. Kusutaki, M. Furukawa, Preconcentration of Pb with aminosilanized Fe3O4 nanopowders in environmental water followed by electrothermal atomic absorption spectrometric determination, Chem. Eng., 3 ( 2019) 74.
W.S.J. Hummers, R.E. Offeman, Preparation of graphitic oxide, J. Am. Chem. Soc., 80 (1958) 1339.
S. Khodabakhshi, F. Marahel, A. Rashidi, M. Khaleghi Abbasabadi, A green synthesis of substituted coumarins using nano graphene oxide as recyclable catalyst, J. Chin. Chem. Soc., 62 (2015) 389-392.
H. Shirkhanloo, M. K. Abbasabadi, F. Hosseini, A. Faghihi, Nanographene oxide modified phenyl methanethiol nanomagnetic composite for rapid separation of aluminum in wastewaters, foods, and vegetable samples by microwave dispersive magnetic micro solid-phase extraction, Food Chem.,347 (2021) 129042.
M.K. Abbasabadi, A. Rashidi, S. Khodabakhshi, Benzenesulfonic acid-grafted graphene as a new and green nanoadsorbent in hydrogen sulfide removal. J. Nat. Gas Sci. Eng., 28 (2016) 87-94.
M. Khaleghi-Abbasabadi, D. Azarifar, Magnetic Fe3O4-supported sulfonic acid-functionalized graphene oxide (Fe3O4@GO-naphthalene-SO3H): a novel and recyclable nanocatalyst for green one-pot synthesis, Res. Chem. Intermed., 45 (2019) 2095-2118.
.
JOURNAL PUBLISHING AGREEMENT
PLEASE PROVIDE US THE FOLLOWING INFORMATION,
Article entitled:
Corresponding author:
To be published in the journal:
Your Status
I am the sole author of the manuscript
- I am an Iranian government employee.
- I am a European government employee
- I am a Asian government
- None of the above
I am one author signing on behalf of all co-authors of the manuscript
- I am an Iranian government employee.
- I am a European government employee
- I am a Asian government
- None of the above
Please tick the above blanks (as appropriate), review the Journal Publishing Agreement, and then sign and date the document in black ink.
Published Journal Article: the author may share a link to the formal publication through the relevant DOI. Additionally theses and dissertations which contain embedded Published Journal Articles as part of the formal submission may be hosted publicly by the awarding institution with a link to the formal publication through the relevant DOI. Any other sharing of Published Journal Articles is by agreement with the publisher only.
Signed: ______________________________________ Name printed: ___________________________________________
Title and Company (if employer representative): _______________________Date: __________________________________