A rapid removal of xylene from air based on nano-activated carbon in the dynamic and static systems and compared to commercial activated carbon before determination by gas chromatography
Volume 5, Issue 03, Pages 19-30, Sep 2022 *** Field: Analytical Method in occupational Health
Abstract
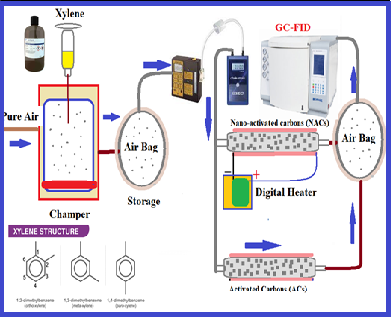
As main air pollutants, volatile organic compounds (VOCs) must be paid special attention. In this study, the removal efficiency of xylene from the air was investigated by nano-activated carbons (NACs) as an efficient adsorbent and compared to commercial activated carbons (ACs). In the chamber, the xylene vapor in pure air was generated, stored in the airbag (5 Li), and moved to adsorbents. Then, the xylene vapor was absorbed on the NAC/AC adsorbents and desorbed from it by a heat accessory. The efficiency of xylene removal with NACs and ACs was investigated in the dynamic and static systems based on 100-700 mg L-1 of xylene, flow rates of 100 ml min-1, and 100 mg of adsorbent at a humidity of 32% (25°C). Xylene concentrations were determined by gas chromatography equipped with a flame ionization detector (GC-FID). In the batch system, the maximum absorption capacity for NACs and ACs was obtained at 205.2 mg g-1 and 116.8 mg g-1, respectively. The mean adsorption efficiency for NACs and ACs adsorbents was obtained at 98.5% and 76.55%, respectively. The RSD% for NACs ranged between 1.1-2.5% in optimized conditions. The characterizations of the NACs adsorbent showed that the particle-size range was between 35-100 nm.
References
ACGIH, TLVs and BEIs, threshold limit values for chemical substances and physical agents and biological exposure indices, Signature Publications, Cincinnati, 2011. https://www.acgih.org/science/tlv-bei-guidelines/
F. Çeçen, Ö. Aktaş, Activated carbon for water and wastewater treatment: integration of adsorption and biological treatment, John Wiley & Sons, 2011. https://doi.org/10.1002/9783527639441.
H.R. Mokarami, Removal of xylene from waste air stream using catalytic ozonation process, Iran. J. health Environ., 3 (2010) 239-250. http://ijhe.tums.ac.ir/article-1-105-en.html
X. Zhang, B. Gao, A.E. Creamer, C. Cao, Y. Li, Adsorption of VOCs onto engineered carbon materials: A review, J. Hazard. Mater., 338 (2017) 102–123. https://doi.org/10.1016/j.jhazmat.2017.05.013.
B. Bina, M.M. Amin, A. Rashidi, H. Pourzamani, Benzene and toluene removal by carbon nanotubes fromaqueous solution, Arch. Environ. Prot., 38 (2012) 3–25. https://doi.org/10.2478/v10265-012-0001-0.
L.H. Quynh Anh, Removal of O,M,P-Xylene from air samples on oxidized carbon nanotubes cartridges, Vietnam J. Sci. Technol., 56 (2018) 226–233. https://doi.org/10.15625/2525-2518/56/2a/12690.
F. Su, C. Lu, S. Hu, Adsorption of benzene, toluene, ethylbenzene and p-xylene by NaOCl-oxidized carbon nanotubes, Colloids Surfaces A Physicochem. Eng. Asp., 353 (2010) 83–91. https://doi.org/10.1016/j.colsurfa.2009.10.025.
M. Jafarizaveh, H. Shirkhanloo, F. Golbabaei, A. Tabrizi, K. Azam, M. Ghasemkhani, Nobel method for xylene removal from air on nano activated carbon adsorbent compared to NIOSH approved carbon adsorbent, J. Health Saf. Work, 6 (2016) 23.
http://jhsw.tums.ac.ir/article-1-5374-en.html
H.Q.A. Le, D.T. Phan, Investigation of BTEX adsorption on carbon nanotubes cartridges from air samples, Appl. Mech. Mater., 889 (2019) 216–222. https://doi.org/10.4028/www.scientific.net/amm.889.216.
H.A. Rangkooy, F. Jahani, A. Siahi Ahangar, Effect of the Type of Ultraviolet on the Photocatalytic Removal of Xylene as a Pollutant in the Air Using TiO2 Nanoparticles Fixed on the activated carbon, J. Occup. Hyg. Eng., 5 (2019) 26–32.
http://johe.umsha.ac.ir/article-1-461-en.html
A.A.M. Beigi, M. Yousefi, M. Abdouss, Room temperature imidazolium-based ionic liquids as scavengers for hydrogen sulfide removal of crude oil, Anal. Methods Environ. Chem. J., 1 (2018) 11–22. https://doi.org/10.24200/amecj.v1.i01.32.
S.M. Mostafavi, A. Ebrahimi, Mercury determination in work place air and human biological samples based on dispersive liquid-liquid micro-extraction coupled with cold vapor atomic absorption spectrometry, Anal. Methods Environ. Chem. J., 2 (2019) 49–58. https://doi.org/10.24200/thamecj.v2.i04.81.
P. Paydar, A.F. Zarandi, Air Pollution Method: A new method based on ionic liquid passed on mesoporous silica nanoparticles for removal of manganese dust in the workplace air, Anal. Methods Environ. Chem. J., 2 (2019) 5–14. https://doi.org/10.24200/amecj.v2.i01.52.
S.A.H. Mirzahosseini, Environmental Health Analysis: Assessing the emission levels of benzene from the fuel tanks doors of the vehicles in Tehran city, Anal. Methods Environ. Chem. J., 2 (2019) 49–54. https://doi.org/10.24200/amecj.v2.i01.47.
C. Jamshidzadeh, H. Shirkhanloo, A new analytical method based on bismuth oxide-fullerene nanoparticles and photocatalytic oxidation technique for toluene removal from workplace air, Anal. Methods Environ. Chem. J., 2 (2019) 73–86. https://doi.org/10.24200/amecj.v2.i01.55.
A. Vahid, Determination of H2S in crude oil via a rapid, reliable and sensitive method, Anal. Methods Environ. Chem. J., 2 (2019) 37–44. https://doi.org/10.24200/amecj.v2.i2.61.
M. Bagheri Hosseinabadi, S. Timoori, A. Faghihi Zarandi, Functionalized graphene-trimethoxyphenyl silane for toluene removal from workplace air by sorbent gas extraction method, Anal. Methods Environ. Chem. J., 2 (2019) 45–54. https://doi.org/10.24200/amecj.v2.i2.63.
A. Ebrahimi, A. Salarifar, Air pollution Analysis: Nickel paste on Multi-walled carbon nanotubes as novel adsorbent for the mercury removal from air, Anal. Methods Environ. Chem. J., 2 (2019) 79–88. https://doi.org/10.24200/amecj.v2.i03.70.
M. Arjomandi, H. Shirkhanloo, A Review: Analytical methods for heavy metals determination in environment and human samples, Anal. Methods Environ. Chem. J., 2 (2019) 97–126. https://doi.org/10.24200/amecj.v2.i03.73.
M. Gou, B.B. Yarahmadi, Removal of ethylbenzene from air by graphene quantum dots and multi wall carbon nanotubes in present of UV radiation, Anal. Methods Environ. Chem. J., 2 (2019) 59–70. https://doi.org/10.24200/amecj.v2.i04.82.
P.M. Eller, M.E. Cassinelli, NIOSH manual of analytical methods, 4th ed, Diane Publishing, 1996. https://doi.org/10.5860/choice.33-2747.
A. Tabrizi, F. Golbabaei, H. Shirkhanloo, M. Jafarizaveh, K. Azam, R. Yarahmadi, Evaluation of the adsorption capacity of nano-graphene and nano-graphene oxide for xylene removal from air and their comparison with the standard adsorbent of activated carbon to introduce the optimized one, J. Heal. Saf. Work, 6 (2016) 25-34. http://jhsw.tums.ac.ir/article-1-5415-en.html
O.G. Apul, Q. Wang, Y. Zhou, T. Karanfil, Adsorption of aromatic organic contaminants by graphene nanosheets: Comparison with carbon nanotubes and activated carbon, Water Res., 47 (2013) 1648–1654. https://doi.org/10.1016/j.watres.2012.12.031.
Z. Vahdat Parast, H. Asilian, A. Jonidi Jafari, Adsorption of xylene from air by natural iranian zeolite, Heal. Scope, 3 (2014). e17528. https://doi.org/10.17795/jhealthscope-17528.
H. Shirkhanloo, M. Osanloo, O.Q. Dadras, Nobel method for toluene removal from air based on ionic liquid modified nano-graphen, Int. J. Occup. Hyg., 6 (2014) 1–5. https://ijoh.tums.ac.ir/index.php/ijoh/article/view/89
Copyright (c) 2022 Analytical Methods in Environmental Chemistry Journal

This work is licensed under a Creative Commons Attribution 4.0 International License.
JOURNAL PUBLISHING AGREEMENT
PLEASE PROVIDE US THE FOLLOWING INFORMATION,
Article entitled:
Corresponding author:
To be published in the journal:
Your Status
I am the sole author of the manuscript
- I am an Iranian government employee.
- I am a European government employee
- I am a Asian government
- None of the above
I am one author signing on behalf of all co-authors of the manuscript
- I am an Iranian government employee.
- I am a European government employee
- I am a Asian government
- None of the above
Please tick the above blanks (as appropriate), review the Journal Publishing Agreement, and then sign and date the document in black ink.
Published Journal Article: the author may share a link to the formal publication through the relevant DOI. Additionally theses and dissertations which contain embedded Published Journal Articles as part of the formal submission may be hosted publicly by the awarding institution with a link to the formal publication through the relevant DOI. Any other sharing of Published Journal Articles is by agreement with the publisher only.
Signed: ______________________________________ Name printed: ___________________________________________
Title and Company (if employer representative): _______________________Date: __________________________________