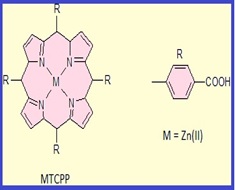
The use of zinc metalloporphyrin grafted magnetic nanoparticles for the removal of sulfate ions from wastewaters
Vol 2, Issue 02, Pages 65-76,*** Field: Water and Wastewater Analysis
Abstract
This study investigates an application of zinc metalloporphyrin grafted Fe3O4 nanoparticles as a new adsorbent for removal of sulfate ions from wastewaters. The modification of magnetite nanoparticles was conducted by 3-aminopropyltriethoxysilane followed by zinc (II) porphyrin in order to enhance the removal of sulfate ions. Fourier Transform Infrared Spectroscopy (FT-IR), X-ray diffraction (XRD), Transmission Electron Microscopy (TEM) and Scanning Electron Microscopy (SEM) was used to characterize the synthesized nano sorbent. The effect of important experimental factors such as pH, contact time, sorbent dosage and some co-existing anions present in aqueous solutions were investigated. Under optimal conditions (i.e. contact time: 30 min, pH: 6.5 and nanosorbents dosage: 100 mg) for a sulfate sample (50 mL, 50 mgL-1 ) the percentage of the extracted sulfate ions was 94.5%. Regeneration of sulfate adsorbed material could be possible by NaOH solution and the modified magnetic nano sorbent exhibited good reusability.
References
A.J. Silva, M.B. Varesche, E. Foresti, M. Zaiat, Sulphate removal from industrial wastewater using a packed-bed anaerobic reactor, Process Biochem., 37 (2002) 927-935.
P.N.L. Lens, A. Visser, A.J.H. Janssen, L.W. Hulshoff Pol, G. Lettinga, Biotechnological Treatment of Sulfate-Rich Wastewaters, Crit. Rev. Environ. Sci. Technol., 28 (1998) 41-88.
C. T. Benatti, C. R. Tavares, E. Lenzi, Sulfate removal from waste chemicals by precipitation, J. Environ. Manag., 90 (2009) 504-511.
R. Ghigliazza, A. Lodi, M. Rovatti, Kinetic and process considerations on biological reduction of soluble and scarcely soluble sulfates, Resour. Conserv. Recy, 29 (2000) 181-194.
World Health Organization (WHO), ISBN 978 92 4 154815 1, Guidelines for Drinking-water Quality, 2011.
F. Register, National Primary and Secondary Drinking Water Regulations; Synthetic Organic Chemicals and Inorganic Chemical, Fed. Reg., 55 (1989) 30-37.
G. Muyzer, A. J. M. Stams, The ecology and biotechnology of sulphate-reducing bacteria, Nat. Rev. Microbiol., 6 (2008) 441-454.
S.Y. Chang, N.Y. Zheng, C.S. Chen, C.D. Chen, Y.Y. Chen, C.R.C. Wang, Analysis of peptides and proteins affinity-bound to iron oxide nanoparticles by MALDI MS, J. Am. Soc. Mass Spectrom., 18 (2007) 910-918.
X. Zhao, Y. Shi, T. Wang, Y. Cai, G. Jiang, Preparation of silica-magnetite nanoparticle mixed hemimicelle sorbents for extraction of several typical phenolic compounds from environmental water samples, J. Chromatogr. A., 1188 (2008) 140-147.
C. Huang, B. Hu, Speciation of inorganic tellurium from seawater by ICP‐MS following magnetic SPE separation and preconcentration, J. Sep. Sci, 31 (2008) 760-767.
J.S. Suleiman, B. Hu, H. Peng, C. Huang, Separation/preconcentration of trace amounts of Cr, Cu and Pb in environmental samples by magnetic solid-phase extraction with Bismuthiol-II-immobilized magnetic nanoparticles and their determination by ICP-OES, Talanta, 77 (2009) 1579-1583.
B. Zargar, H. Parham, A. Hatamie, Modified iron oxide nanoparticles as solid phase extractor for spectrophotometeric determination and separation of basic fuchsin, Talanta, 77 (2009) 1328-1331.
L. Sun, L. Chen, X. Sun, X. Du, Y. Yu, D. He, H. Xu, Q. Zeng, H. Wang, L. Ding, Analysis of sulfonamides in environmental water samples based on magnetic mixed hemimicelles solid-phase extraction coupled with HPLC–UV detection, Chemosphere, 77 (2009) 1306-1312.
H. Parham, N. Rahbar, Solid phase extraction–spectrophotometric determination of salicylic acid using magnetic iron oxide nanoparticles as extractor, J. Pharm. Biomed. Anal., 50 (2009) 58-63.
L. Sun, C. Zhang, L. Chen, J. Liu, H. Jin, H. Xu, L. Ding, Preparation of alumina-coated magnetite nanoparticle for extraction of trimethoprim from environmental water samples based on mixed hemimicelles solid-phase extraction, Anal. Chim. Acta, 638 (2009) 162-168.
M. Faraji, Y. Yamini, A. Saleh, M. Rezaee, M. Ghambarian, R. Hassani, A nanoparticle-based solid-phase extraction procedure followed by flow injection inductively coupled plasma-optical emission spectrometry to determine some heavy metal ions in water samples, Anal. Chim. Acta, 659 (2010) 172-177.
K. Moeller, J. Kobler, T. Bein, Colloidal Suspensions of Nanometer‐Sized Mesoporous Silica, Adv. Funct. Mater., 17 (2007) 605-612.
K.J. Klabunde, Nanoscale Material in Chemistry, Wiley-Interscience, New York, 2001.
M. Faraji, Y. Yamini, M. Rezaee, Magnetic nanoparticles: Synthesis, stabilization, functionalization, characterization, and applications, J. Iran. Chem. Soc., 7 (2010) 1-37.
Ch. Huang, B. Hu, Silica-coated magnetic nanoparticles modified with γ-mercaptopropyltrimethoxysilane for fast and selective solid phase extraction of trace amounts of Cd, Cu, Hg, and Pb in environmental and biological samples prior to their determination by inductively coupled plasma mass spectrometry, Spectrochim. Acta Part B, 6 (2008) 437-444.
K. Jainae, K. Sanuwong, J. Nuangjamnong, N. Sukpirom, F. Unob, Extraction and recovery of precious metal ions in wastewater by polystyrene-coated magnetic particles functionalized with 2-(3-(2-aminoethylthio)propylthio)ethanamine, Chem. Eng. J., 160 (2010) 586-593.
Zh. Yong-Gang, Sh. Hao-Yu, P. Sheng-Dong, H. Mei-Qin, J. Hazard. Mater., 182 (2010) 295.
X. Wang, L.Wang, X. He, Y. Zhang, L. Chen, A molecularly imprinted polymer-coated nanocomposite of magnetic nanoparticles for estrone recognition, Talanta, 7 (2009) 327-332.
.P.D Beer, .PA. Gale, Anion Recognition and Sensing: The State of the Art and Future Perspectives, Chem. Int. Edu., 40 (2001) 486-516.
A. Bianchi, K. Bowman-James, E. Garcia-Espan., Supramolecular Chemistry of Anions, Wiley/VCH, 1997.
J.T. Mitchell-Koch, M. Pietrzak, E. Malinowska, M. E. Meyerhoff, Aluminum(III) Porphyrins as Ionophores for Fluoride Selective Polymeric Membrane Electrodes, Electroanal., 18 (2006) 551-557 .
T. Poursaberi, M. Hassanisadi, K. Torkestani, M. Zare, Development of zirconium (IV)-metalloporphyrin grafted Fe3O4 nanoparticles for efficient fluoride removal, Chem. Eng. J, 189 (2012) 117-125.
T. Poursaberi, M. Karimi, M. Hassanisadi, H. Sereshti, Magnetic removal of nitrate ions from aqueous solution using amino-silica coated magnetic nanoparticles modified by oxovanadium(IV) porphyrin, J. Porphyrins Phthalocyanines, 17 (2013) 359-366.
J. G. Erdman, V. G. Ramsey, N.W. Kalenda, W. E. Hanson, Synthesis and Properties of Porphyrin Vanadium Complexes, J. Am. Chem. Soc., 78 (1956) 5844-5847.
X.L. Zhao, Y.L. Shi, Y.Q. Cai, S.F. Mou, Cetyltrimethylammonium Bromide-Coated Magnetic Nanoparticles for the Preconcentration of Phenolic Compounds from Environmental Water Samples, Environ. Sci. Technol., 42 (2008) 1201-1206.
R.B.S. Merrifield, J. Am. Chem. Soc. 2149 (1963) 85.
V.V.Egorov, E.M.Rakhman’ko, A.A.Rat’ko, Metalloporphyrin-Based Anion-Selective Electrodes with Unusual Selectivity, J. Anal. Chem., 57 (2002) 46-53.
K. M. Morehouset and P. Neta, J. Phys. Chem., 18 (1984) 88.
Y. Kang, C. Lutz, S.A. Hong, D. Sung, J.S. Lee, J.H. Shin, H. Nam, G.S. Cha, M.E. Meyerhoff, Development of a Fluoride-Selective Electrode based on Scandium(III) Octaethylporphyrin in a Plasticized Polymeric Membrane, Bull. Korean Chem. Soc., 31 (2010) 17-29.
Copyright (c) 2019 Analytical Methods in Environmental Chemistry Journal

This work is licensed under a Creative Commons Attribution 4.0 International License.
JOURNAL PUBLISHING AGREEMENT
PLEASE PROVIDE US THE FOLLOWING INFORMATION,
Article entitled:
Corresponding author:
To be published in the journal:
Your Status
I am the sole author of the manuscript
- I am an Iranian government employee.
- I am a European government employee
- I am a Asian government
- None of the above
I am one author signing on behalf of all co-authors of the manuscript
- I am an Iranian government employee.
- I am a European government employee
- I am a Asian government
- None of the above
Please tick the above blanks (as appropriate), review the Journal Publishing Agreement, and then sign and date the document in black ink.
Published Journal Article: the author may share a link to the formal publication through the relevant DOI. Additionally theses and dissertations which contain embedded Published Journal Articles as part of the formal submission may be hosted publicly by the awarding institution with a link to the formal publication through the relevant DOI. Any other sharing of Published Journal Articles is by agreement with the publisher only.
Signed: ______________________________________ Name printed: ___________________________________________
Title and Company (if employer representative): _______________________Date: __________________________________