Reusable and sustainable graphene oxide/metal–organic framework-74/Fe3O4/polytyramine nanocomposite for simultaneous trace level quantification of five fluoroquinolones in egg samples by high performance liquid chromatography
Volume 4, Issue 02, Pages 5-24, Jun 2021 *** Field: Analytical Nano Chemistry
Abstract
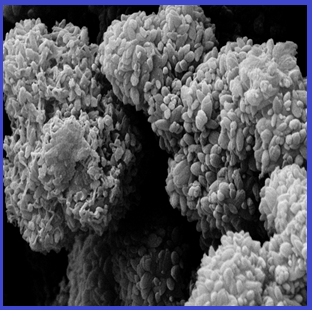
A nanohybrid material termed graphene oxide/metal-organic framework-74/Fe3O4/polytyramine (GO/MOF-74/Fe3O4/PTy) was fabricated and applied in magnetic dispersive micro-solid phase extraction (MD-µ-SPE) coupled with high performance liquid chromatography (HPLC) for simultaneous determination of fluoroquinolones compounds including, ofloxacin, ciprofloxacin, lomefloxacin, enrofloxacin and sperfloxacin in egg samples. The GO/MOF-74/Fe3O4/PTy nanocomposite was fabricated through an in situ synthesis of MOF-74 in the presence of magnetic GO and followed with an oxidative polymerization of tyramine using horsedish peroxide (HRP) enzyme. The modifier agents improved the merits of the nanoporous sorbent. Extraction protocols based on GO/MOF nanocomposites have various benefit such as, the high stability, the tunable porosity, the fast mast transfer and reasonable enrichment factor. The fabricated material was characterized via energy dispersive x-ray analysis (EDX), the scanning electron microscopy (SEM), Fourier-transform infrared spectroscopy (FT-IR), and the x-ray diffraction (XRD). The calibration curves revealed linearity (0.992 ≤ r2 ≤ 0.997) in the ranges of 1.0-475.0, 0.5-350.0, 0.5-350.0, 0.5-375.0 and 1.5-300.0 ng mL-1 with limit of detections (LODs, S/N=3) of 0.3, 0.1, 0.2, 0.1 and 0.4 ng mL-1 for ofloxacin, ciprofloxacin, lomefloxacin, enrofloxacin and sperfloxacin, respectively. The intra-assay (≤7.7%, n = 9) and inter-assay (≤7.0%, n = 9) precisions along with accuracy less than 9.0% were obtained.
References
H. Wu, Y. Liu, J. Chang, B. Zhao, Y. Huo, Zh. Wang, Y. Shi, Extraction of Five Fluoroquinolones in Eggs by Magnetic Solid-Phase Extraction with Fe3O4–MoS2 and Determination by HPLC-UV, Food Anal. Methods, 12 (2019)712-721.
Wm. Scheld, Maintaining fluoroquinolone class efficacy: review of Influencing factors, Emerg. Infect. Dis., 9 (2003)1-9.
H. Yan, F. Qiao, KH. Row, Molecularly imprinted-matrix solid-phase dispersion for selective extraction of five fluoroquinolones in eggs and tissue, Anal. Chem., 79 (2007) 8242-8248.
A. Gajda, A. Posyniak, J. Zmudzki, M. Gbylik, T. Bladek, Determination of (fluoro)quinolones in eggs by liquid chromatography with fluorescence detection and confirmation by liquid chromatography–tandem mass spectrometry, food chem., 135 (2012) 430-439.
JF. Huang,B. Lin, QW. Yu, YQ. Feng, Determination of fluoroquinolones in eggs using in-tubensolid-phase microextraction coupled to high-performance liquid chromatography, Anal. Bioanal. Chem., 384 (2006) 1228–1235.
M. Sturini, A. Speltini, L. Pretali, E. Fasani, A. Profumo, Solid-phase extraction and HPLC determination of fluoroquinolones in surface waters, J. Sep. Sci., 32 (2009) 3020-3028.
IS. Ibarra, JA. Rodriguez, CA. Galán-Vidal, A. Cepeda, JA. Miranda, Magnetic solid phase extraction applied to food analysis, J. Chem., 8 (2015) 1-13.
ShV. Gopalan, AN. Hasanah, MI-SPE, M-SPE AND M-SPD recent application on solid phase extraction for compound extraction of complex matrices, Int. J. App. Pharm., 11 (2019)16-25.
L. Reis, L. Lorena Vidal, A. Canals, Determination of siloxanes in water samples employing graphene oxide/Fe3O4 nanocomposite as sorbent for magnetic solid-phase extraction prior to gas chromatography-mass spectrometry, J. Sci. Food Agric., 41 (2018) 4177-4184.
A. Issa, Kh. Al‐Saad, A. S. Luyt, Magnetic solid phase extraction for chromatographic separation of carbamates, J. Sci. Food Agric., 101 (2017) 2038-2049.
T. Khezeli, A. Daneshfar, Development of dispersive micro-solid phase extraction based on micro and nano sorbents, Trends Anal. Chem., 89 (2017)99-118.
M. Zeeb, H. Farahani, Graphene oxide/Fe3O4@polythionine nanocomposite as an efficient sorbent for magnetic solid-phase extraction followed by high-performance liquid chromatography for the determination of duloxetine in human plasma, Chem. Paper, 72 (2018) 15-27.
N. Li, HL. Jiang, X. Wang, X. Wang, Gu. Xu, B. Zhang, L. Wang, RS. Zhao, JM. Lin, Recent advances in graphene-based magnetic composites for magnetic solid-phase extraction, Trends Anal. Chem., 102 (2018) 60-74.
A. Roostaie, Mohammadiazar, H. Bargozin, S. Ehteshami, A modified nanoporous silica aerogel as a new sorbent for needle trap extraction of chlorobenzenes from water samples, Chromatogra., 81 (2018) 649-655.
Ch. Xu, X. Wang, J. Zhu, Graphene-metal particle nanocomposites, J. Phys. Chem. C, 112 (2008) 19841–19845.
NM. El-Shafai, ME. El-Khouly, M. El-Kemary, MS. Ramadana, MS. Masoud, Graphene oxide–metal oxide nanocomposites: fabrication, characterization and removal of cationic rhodamine B dye, RSC Adv., 8 (2018) 13323-13332.
Xu. XubiaoLuo, Ch. Wang, Sh. Luo, R. Dong, X. Tu, Gu. Zeng, Adsorption of As (III) and As (V) from water using magnetite Fe3O4-reduced graphite oxide MnO2 nanocomposites, Chem. Eng. J., 187 (2012) 45-52.
WK. Chee, H. Lim, HN. Ming, I. Harrison, Nanocomposites of graphene/polymers: a review, RSC Adv., 5 (2015) 68014-68051.
F. Xu, Y. Yu, J. Yan, Q. Xia, H. Wang, J. Li, Zh. Li, Ultrafast room temperature synthesis of GrO@HKUST-1 composites with high CO2 adsorption capacity and CO2/N2 adsorption selectivity, Chem. Eng. J., 303 (2016) 231-237.
A. Amiri, F. Ghaemi, B. Maleki, Hybrid nanocomposites prepared from a metal-organic framework of type MOF-199(Cu) and graphene or fullerene as sorbents for dispersive solid phase extraction of polycyclic aromatic hydrocarbons, Microchim. Acta, 3 (2019) 131-139.
I. Ahmed, NA. Khan, SH. Jhung, Graphite oxide/metal−organic framework (MIL-101): remarkable performance in the adsorptive denitrogenation of model fuels, Inorg. Chem., 24 (2013)14155-14161.
JR. Li, RJ. Kuppler, HC. Zhou, Selective gas adsorption and separation in metal–organic frameworks, Chem. Soc. Rev., 38 (2009) 1477-1504.
J. An, SJ. Geib, NL. Rosi, High and selective CO2 uptake in a cobalt adeninate metal−organic framework exhibiting pyrimidine- and amino-decorated pores, Am. Chem. Soc., 132 (2010) 38-39.
D. Britt, H. Furukawa, B. Wang, TG. Glover, OM. Yaghi, Highly efficient separation of carbon dioxide by a metal-organic framework replete with open metal sites, Nat. Acad. Sci. U. S. A., 106 (2009) 20637-20640.
YS. Bae, AM. Spokoyny, OK. Farha, RQ. Snurr, JT. Hupp, CA. Mirkin, Separation of gas mixtures using Co(II) carborane-based porous coordination polymers, Chem. Commun., 46 (2010) 3478-3480.
H. Li, K. Wang, Y. Sun, ChT. Lollar, J. Li, HC. Zhou, Recent advances in gas storage and separation using metal–organic frameworks, Mater. today, 21 (2018) 108-121.
H. Li, L. Li, RB. Lin, W. Zhou, Zh. Zhang, Sh. Xiang, B. Chen, Porous metal-organic frameworks for gas storage and separation: Status and challenges, Energy Chem., 1 (2019) 100006-100049.
A. Chowdhury, The applications of metal-organic-frameworks in controlled release of drugs, Rev. J. Chem., 7 (2017) 1-22.
H. Zhou, X. Liu, J. Zhang, X. Yan, Y. Liu, Yua, Enhanced room-temperature hydrogen storage capacity in Pt-loaded graphene oxide/HKUST-1composites, Int. J. Hydrog. Energy, 39 (2014) 2160-2167.
X. Liu, H. Zhou, Y. Zhang, Y. Liu, A. Yuan, Syntheses, Characterizations and adsorption properties of MIL‐101/graphene oxide composites, Chin. J. Chem., 30 (2012) 2563-2566.
LCh. Lin, D. Paik, J. Kim, Understanding gas adsorption in MOF-5/graphene oxide composite materials, Phys. Chem. Chem. Phys., 19 (2017) 11639-11644.
P. Peipei Yang P, Q. Liu, J. Liu, H. Zhang, Zh. Li, R. Li, L. Liu, J. Wang, Interfacial growth of metal organic framework (UiO-66) on the functionalization of graphene oxide (GO) as a suitable seawater sorbent for extraction of uranium(VI), J. Mater. Chem. A, 5 (2017) 17933-17942.
C. Petit, TJ. Bandosz, Synthesis, Characterization, and ammonia adsorption properties of mesoporous metal–organic framework (MIL(Fe))–graphite oxide composites: exploring the limits of materials fabrication, Adv. Funct. Mater., 21 (2011) 2108–2117.
Ad. Oliveira, GFd. Lima, HAD. Abreu, Structural and electronic properties of M-MOF-74 (M = Mg, Co or Mn), Chem. Phys. Lett., 691 (2018) 283–290.
M. Díaz-García, A. Mayoral, I. Díaz, M. Sánchez-Sánchez, Nanoscaled M‑MOF-74 materials prepared at room temperature, Cryst. Growth Des., 14 (2014) 2479−2487.
TG. Glover, GW. Peterson, BJ. Schindler, D. Britt, O. Yaghi, MOF-74 building unit has a direct impact on toxic gas adsorption, Chem. Eng. Sci., 66 (2011) 163-170.
S. Patra, E. Roy, R. Madhuri, PK. Sharma, Fast selective preconcentration of europium from wastewater and coal soil by graphene oxide/silane@Fe3O4 dendritic nanostructure, Sci. Technol., 49 (2015) 6117-6126.
Ö Metin, S. Aydoğan, K. Meral, A new route for the synthesis of graphene oxide-Fe3O4 (GO–Fe3O4) nanocomposites and their schottky diode applications, J. Alloy Compd., 585 (2013) 681-688.
BS. Rodríguez, JH. Borges, AV. Herrera-Herrera, M. Rodríguez-Delgado M, Multiresidue analysis of oestrogenic compounds in cow, goat, sheep and human milk using core-shell polydopamine coated magnetic nanoparticles as extraction sorbent in micro-dispersive solid-phase extraction followed by ultra-high-performance liquid chromatography tandem mass spectrometry, Anal. Bioanal. Chem., 410 (2018) 2031-2042.
A. Mehdinia, N. Khodaee, A. Jabbari, Fabrication of graphene/Fe3O4@polythiophene nanocomposite and its application in the magnetic solid-phase extraction of polycyclic aromatic hydrocarbons from environmental water samples, Anal. Chim. Acta, 868 (2015) 1–9.
GR. Lopes, DC. Pinto, AMS. Silva, Horseradish peroxidase (HRP) as a tool in green chemistry, RSC Adv., 4 (2014) 37244-37265.
NC.Veitch, Horseradish peroxidase: a modern view of a classic enzyme. Phytochem., 65 (2004) 249–259.
H. Kawakita, K. Hamamoto, K. Ohto, Kb. Inoue, Polyphenol polymerization by horseradish peroxidase for metal adsorption studies, Ind. Eng. Chem. Res., 48 (2009) 4440–4444.
F. Pourbahman, M. Zeeb, A. Monzavi, SS. Homami, Simultaneous trace monitoring of prokinetic drugs in human plasma using magnetic dispersive micro-solid phase extraction based on a new graphene oxide/ metal–organic framework-74/Fe3O4/ polytyramine nanoporous composite in combination with HPLC, Chem. Papers, 73 (2019) 3135-3150.
J. Song, X. Wang, ChT. Chang ChT, Preparation and characterization of graphene oxide, J. nanometer., 4 (2014) 1-6.
SK. Kimitoshi, JG. Li, H. Kamiya, T. Ishigaki, Ultrasonic dispersion of TiO2 nanoparticles in aqueous suspension, J. Am. Ceram., Soc., 91 (2008) 2481–2487.
AK. Adhikari, K. Lin, Synthesis, fine structural characterization, and CO, Nanosci. Nanotech., 13 (2013) 1–9.
DW. Wang, YQ. Li, QH. Wang, Nanostructured Fe2O3–graphene composite as a novel electrode material for supercapacitors, J. Solid State Electrochem., 16 (2012) 2095–2102
Zh. Li, B. Yu, H. Cong, H. Hua Yuan, Q. Peng, Recent development and application of solid phase extraction materials, Rev. Adv. Mater. Sci., 48 (2017) 87-111.
E. Tahmasebi, MY. Masoomi, Y. Yamini, A. Morsali, Application of a Zn(II) based metal-organic framework as an efficient solid-phase extraction sorbent for preconcentration of plasticizer compounds, J. Name., 00 (2013) 1-3.
M.A. Farajzadeh, A. Yadeghari, M. Abbaspour, Dispersive solid phase extraction using magnetic nanoparticles performed in a narrow-bored tube for extraction of atorvastatin, losartan, and valsartan in plasma, Adv. Pharm. Bull., 9 (2019) 138-146.
A. babaeia, M. Zeeb, A. Es-haghi, Magnetic dispersive solid-phase extraction based on graphene oxide/Fe3O4@polythionine nanocomposite followed by atomic absorption spectrometry for zinc monitoring in water, flour,
celery and egg, J. Sci. Food Agric., 98 (2018) 3571-3579.
JH. Shim, MH. Lee, MR. Kim, CJ. Lee, IS. Kim, Simultaneous measurement of fluoroquinolones in eggs by a combination of supercritical fluid extraction and high pressure liquid chromatography, Biosci. Biotechnol. Biochem., 67 (2014) 1342–1348.
V. Jimenez, R. Companyo, J. Guiteras, Validation of a method for the analysis of nine quinolones in eggs by pressurized liquid extraction and liquid chromatography with fluorescence detection, Talanta, 85 (2011) 596–606.
Copyright (c) 2021 Analytical Methods in Environmental Chemistry Journal

This work is licensed under a Creative Commons Attribution 4.0 International License.
JOURNAL PUBLISHING AGREEMENT
PLEASE PROVIDE US THE FOLLOWING INFORMATION,
Article entitled:
Corresponding author:
To be published in the journal:
Your Status
I am the sole author of the manuscript
- I am an Iranian government employee.
- I am a European government employee
- I am a Asian government
- None of the above
I am one author signing on behalf of all co-authors of the manuscript
- I am an Iranian government employee.
- I am a European government employee
- I am a Asian government
- None of the above
Please tick the above blanks (as appropriate), review the Journal Publishing Agreement, and then sign and date the document in black ink.
Published Journal Article: the author may share a link to the formal publication through the relevant DOI. Additionally theses and dissertations which contain embedded Published Journal Articles as part of the formal submission may be hosted publicly by the awarding institution with a link to the formal publication through the relevant DOI. Any other sharing of Published Journal Articles is by agreement with the publisher only.
Signed: ______________________________________ Name printed: ___________________________________________
Title and Company (if employer representative): _______________________Date: __________________________________