A rapid cadmium determination based on ion selective membrane potentiometric sensor by bis (salicylaldehydo)ethylenediimine as carrier
Volume 4, Issue 02, Pages 25-33, Jun 2021 *** Field: Analytical Chemistry
Abstract
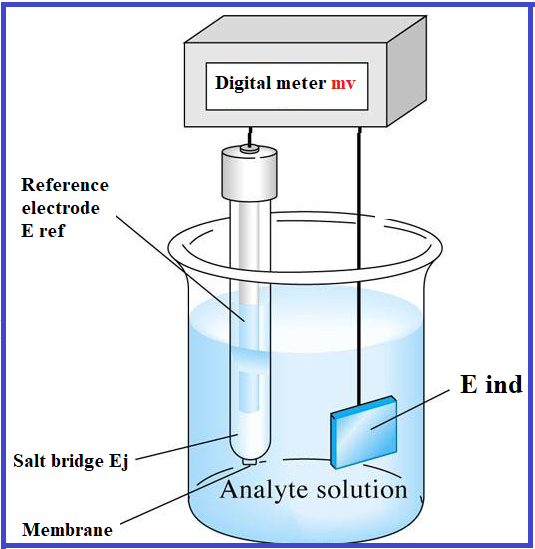
An ion selective potentiometric electrode (IPE) was prepared based on salen material (bis(salicylaldehydo)ethylenediimine) as a suitable carrier for determination of cadmium ions. An acceptable response for cadmium ions obtained over a linear range 8 × 10−7 to 1.0 × 10−2 M with a slope of 29.8 ± 0.8 mV per decade of activity and a detection limit of 3.2 × 10−7 M for Cd (II) ions in water and liquid samples. It has a response time less than 10 s and can be used for at least 2.5 months without any measurable divergence in potential. The ion selective electrode can be used based on potential and potential changes in the pH range 3.5 to 6.5, so, the cadmium determination was obtained at independent pH. Moreover, the selectivity of proposed method in presence of interference ions was studied. The results showed that the other cations do not interfere significantly in response electrode at optimized pH. This electrode was successfully used for the determination of cadmium ions in aqueous samples. The validation was obtained based on ICP analyzer and certified reference material in water samples (CRM, NIST).
References
F. Zheng, B. Hu, Thermo-responsive polymer coated fiber-in-tube capillary microextraction and its application to on-line determination of Co, Ni and Cd by inductively coupled plasma mass spectrometry (ICP-MS), Talanta, 85 (2011) 1166-1173.
P. Wu, Ch. Li, J. Chen, Ch. Zheng, X. Hou, Determination of cadmium in biological samples: An update from 2006 to 2011, Appl. Spectrosc. Rev., 47 (2012) 327-370.
G. Adani, T. Filippini, Dietary intake of acrylamide and risk of breast, endometrial and ovarian cancers: A systematic review and dose-response meta-analysis, Cancer Epidemiol. Biomarkers Prev., 29 (2020) 1095-1106.
L.A. Meira, D.F. de Souza, Application of constrained mixture design and Doehlert matrix in the optimization of dispersive liquid-liquid microextraction assisted by ultrasound for preconcentration and determination of cadmium in sediment and water samples by FAAS, Microchem. J., 130 (2017) 56–63.
M. Naghizadeh, M.A. Taher, M. Behzadi, F.H. Moghaddam, Preparation a novel magnetic natural nano zeolite for preconcentration of cadmium and its determination by ETAAS, Environ. Nanotechnol. Monit. Manag., 8 (2017) 261–267.
J. Zhang, J. Fang, X. Duan, Determination of cadmium in water samples by fast pyrolysis–chemical vapor generation atomic fluorescence spectrometry, Spectrochim. Acta Part B , 122 (2016) 52–55.
M.R. Ganjali, H. Khoshsafar, F. Faridbod, A. Shirzadmehr, M. Javanbakht, P. Norouzi, Room temperature ionic liquids (RTILs) and multiwalled carbon nanotubes (MWCNTs) as modifiers for improvement of carbon paste ion selective electrode response; a comparison study with PVC membrane, Electroanal. Int. J., 21(2009) 2175–98.
R. Yan, Sh. Qiu, L. Tong, Y. Qian, Review of progresses on clinical applications of ion selective electrodes for electrolytic ion tests: from conventional ISEs to graphene-based ISEs, Chem. Speciat. Bioavailab., 28 (2016) 72-77.
W.A. Zoubi, N.A. Mohanna, Membrane sensors based on Schiff bases as chelating ionophores - A review, Spectrochim. Acta A Mol. Biomol. Spectrosc., 132 (2014) 854-870.
C. Mohan, K. Sharma, S. Chandra, Cd(II) ion-selective electrode based on 2–acetylthiophene semicarbazone in polymeric membrane, Anal. Bioanal. Electrochem., 9 (2017) 35-46.
A. Xavier1, N. Srividhya, Synthesis and study of Schiff base ligands, IOSR-J. Appl. Chem., 7 (2014) 6-15.
C. Cretu, L. Caseh, B.J. Tang, V. Badea, E.I. Szerb, G. Mehi, S. Shova, O. Cosistor, Mononuclear Cu(II) complexes of novel salicylidene Schiff bases: synthesis and mesogenic properties, Liq. Cryst., 42 (2015) 1139-1147.
A. Vijayalakshmi, J. Thamarai Selvi, Calcium ion selective electrode based on Schiff base as an electro active material-Its preparation and analytical application, Int. J. Curr. Res., 5 (2013) 2176-2178.
Y.H. Ma, R. Yuan, Y.Q. Chai, X. Wu, W. Zhou, X.L. Liu, F. Deng, New Ni(II) ion-selective electrode based on the N-S Schiff base ligand as neutral carrier in PVC matrix, Anal. Lett., 42 (2009) 2411-2429.
S. Suman, R. Sighn, Thiophene-based Schiff base ligand as ionophore for Ni(II)-selective polyvinyl chloride membrane electrode, J. Polym. Eng., 40 (2020) 481–485.
T. Tamoradi, H. Goudarziafshar, S. Rashki, F. Katouzian, F. Chalabian, Synthesis of new Schiff base ligand and its complexes in the presence of some
transition metal ion and evaluation of their antibacterial properties, Med. Lab. Sci., 11 (2017) 5-10.
K.R. Bandi, A.K. Singh, A. Upadhyay, Biologically active Schiff bases as potentiometric sensor for the selective determination of Nd3+ ion, Electrochim. Acta, 105 (2013) 654-664.
A.Q. Alorabi, M. Abdelbaset, S.A. Zabin, Colorimetric detection of multiple metal ions using Schiff base 1-(2-Thiophenylimino)-4-(N-dimethyl)benzene, Chemosensors, 8 (2020) 1-10.
H.M. Abu Shawish, N. Abu Ghalwa, A.R. Al-Dalou, F.R. Zaggout, S.M. Saadeh, A.A. Abou Assi, Effect of plasticizers and ion-exchangers on the detection limit of tramadol-PVC membrane electrodes, Eurasian J. Anal. Chem., 6 (2011) 70–83.
P. Adhikari, L. Alderson, F. Bender, A.J. Ricco, F. Josse, Investigation of polymer-plasticizer blends as SH-SAW sensor coatings for detection of benzene in water with high sensitivity and long-term stability, ACS Sens., 2 (2017) 157-164.
C. Carey, Plasticizer effects in the PVC membrane of the dibasic phosphate selective electrode, Chemosensors, 3 (2015) 284-294.
A.R. Fakhari, M. Shamsipour, Kh. Ghanbari, Zn(II)-selective membrane electrode based on tetra(2-aminophenyl) porphyrin, Anal. Chim. Acta, 460 (2002) 177–183.
A.R. Fakhari, M. Alaghemand, M. Shamsipur, Iron(III)-selective membrane potentiometric sensor based on 5,10,15,20-tetrakis(pentafluorophenyl)-21H,23H-porphyrin, Anal. Lett., 34 (2001) 1097–1106.
A.R. Fakhari, T. Ahmad Raji, H. Naeimi, Copper-selective PVC membrane electrodes based on salens as carriers, Sens. Actuators B Chem., 104 (2005) 317–323.
Copyright (c) 2021 Analytical Methods in Environmental Chemistry Journal

This work is licensed under a Creative Commons Attribution 4.0 International License.
JOURNAL PUBLISHING AGREEMENT
PLEASE PROVIDE US THE FOLLOWING INFORMATION,
Article entitled:
Corresponding author:
To be published in the journal:
Your Status
I am the sole author of the manuscript
- I am an Iranian government employee.
- I am a European government employee
- I am a Asian government
- None of the above
I am one author signing on behalf of all co-authors of the manuscript
- I am an Iranian government employee.
- I am a European government employee
- I am a Asian government
- None of the above
Please tick the above blanks (as appropriate), review the Journal Publishing Agreement, and then sign and date the document in black ink.
Published Journal Article: the author may share a link to the formal publication through the relevant DOI. Additionally theses and dissertations which contain embedded Published Journal Articles as part of the formal submission may be hosted publicly by the awarding institution with a link to the formal publication through the relevant DOI. Any other sharing of Published Journal Articles is by agreement with the publisher only.
Signed: ______________________________________ Name printed: ___________________________________________
Title and Company (if employer representative): _______________________Date: __________________________________