Determination of fenthion in environmental water samples by dispersive liquid–liquid microextraction coupled with spectrofluorimetric and chemometrics methods
Volume 4, Issue 02, Pages 86-98, Jun 2021 *** Field: Analytical Environmental Chemistry
Abstract
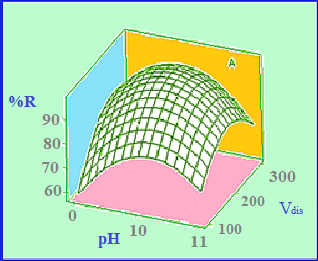
In the present study, a simple, rapid and efficient dispersive liquid–liquid microextraction (DLLME) coupled with spectrofluorimetry and chemometrics methods have been proposed for the preconcentration and determination of fenthion in water samples. Box–Behnken design was applied for multivariate optimization of the extraction conditions (sample pH, the volume of dispersive solvent and volume of extraction solvent). Analysis of variance was performed to study the statistical significance of the variables, their interactions and the model. Under the optimum conditions, the calibration graph was linear in the range of 5.0–110.0 ng mL-1 with the detection limit of 1.23 ng mL-1 (3Sb/m). Parallel factor analysis (PARAFAC) and partial least square (PLS) modelling were applied for the multivariate calibration of the spectrofluorimetric data. The orthogonal signal correction (OSC) was applied for preprocessing of data matrices and the prediction results of model, and the analysis results were statistically compared. The accuracy of the methods, evaluated by the root mean square error of prediction (RMSEP) for fenthion by OSC-PARAFAC and OSC-PLS models were 0.37 and 0.78, respectively. The proposed procedure could be successfully applied for the determination of fenthion in water samples.
References
O. Espinoza-Navarro, C. Ponce-LaRosa, E. Bustos-Obregón, Organophosphorous pesticides: their effects on biosentinel species and humans, control and application in Chile, Int. J. Morphol., 35 (2017) 1069–1074.
K. Vala Ragnarsdottir, Environmental fate and toxicology of organophosphate pesticides, J. Geol. Soc., 157 (2000) 859–876.
P. Sánchez Lizardi, M. K. O’Rourke, R. J. Morris, The effects of organophosphate pesticide exposure on hispanic children’s cognitive and behavioral functioning, J. Pediatr. Psychol., 33 (2007) 91–101.
A. S. H. Derbalah, H. Wakatsuki, T. Yamazaki, H. Sakugawa, Photodegradation kinetics of fenitrothion in various aqueous media and its effect on steroid hormones biosynthesis, Geochem. J., 38 (2004) 201–213.
R. A. Cheke, Effects of the organophosphate fenthion for control of the redbilled quelea Quelea quelea on cholinesterase and haemoglobin concentrations in the blood of target and non-target birds, Ecotoxicol., 21(2012) 1761–1770.
T. Galeano Díaz, A. Guiberteau Cabanillas, M. D. López Soto, J. M. Ortiz, Determination of fenthion and fenthion-sulfoxide, in olive oil and in river water, by square-wave adsorptive-stripping voltammetry, Talanta, 76 (2008) 809–814.
Y. Zhou, An experimental design approach using response surface techniques to obtain optimal liquid chromatography and mass spectrometry conditions to determine the alkaloids in Meconopsi species, J. Chromatogr. A, 1216 (2009) 7013–7023.
S. Boulanouar, S. Mezzache, A. Combès, V. Pichon, Molecularly imprinted polymers for the determination of organophosphorus pesticides in complex samples, Talanta, 176 (2018) 465–478.
T.-K. Ly, T.-D. Ho, P. Behra, Determination of 400 pesticide residues in green tea leaves by UPLC-MS/MS and GC-MS/MS combined with QuEChERS extraction and mixed-mode SPE clean-up method, Food Chemistry, 326 (2020) 126928.
F. Tokay, S. Bağdat, Solid phase extraction and preconcentration of some metal ions using Schiff base immobilised silica gel followed by ICP-OES, Int. J. Environ. Anal. Chem., 00 (2019)1–12.
P. Sun, Y. L. Gao, C. Xu, Y. F. Lian, Determination of six organophosphorus pesticides in water samples by three-dimensional graphene aerogel-based solid-phase extraction combined with gas chromatography/mass spectrometry, RSC Adv., 8 (2018) 10277–10283.
X. C. Huang, J. K. Ma, R. X. Feng, S. L. Wei, Simultaneous determination of five organophosphorus pesticide residues in different food samples by solid-phase microextraction fibers coupled with high-performance liquid chromatography, J. Sci. Food Agric., 99 (2019) 6998–7007.
S. Manafi Khoshmanesh, H. Hamishehkar, H. Razmi, Trace analysis of organophosphorus pesticide residues in fruit juices and vegetables by an electrochemically fabricated solid-phase microextraction fiber coated with a layer-by-layer graphenized graphite/graphene oxide/polyaniline nanocomposite, Anal. Methods, 12 (2020) 3268–3276.
N. S. Pano-Farias, S. G. Ceballos-Magaña, R. Muñiz-Valencia, J. M. Jurado, Á. Alcázar, I. A. Aguayo-Villarreal, Direct immersion single drop micro-extraction method for multi-class pesticides analysis in mango using GC–MS, Food Chem., 237 (2017) 30–38.
A. Niazi, N. Khorshidi, P. Ghaemmaghami, “Microwave-assisted of dispersive liquid-liquid microextraction and spectrophotometric determination of uranium after optimization based on Box-Behnken design and chemometrics methods, Spectrochim. Acta - Part A: Mol. Biomol. Spect., 135 (2015) 69–75.
F. Assadian, A. Niazi, Application of response surface modeling and chemometrics methods for the determination of ofloxacin in human urine using dispersive liquid-liquid microextraction combined with spectrofluorimetry, J. Brazilian Chem. Soc., 28 (2017) 2291–2300.
A. Niazi, S. Habibi, M. Ramezani, Spectrophotometric determination of bismuth in water samples by dispersive liquid-liquid microextraction after multivariate optimization based on box-behnken, J. Chilean Chem. Soc., 58 (2013) 1899–1901.
L. Mousavi, Z.Tamiji, MR. Khoshayand,Applications and opportunities of experimental design for the dispersive liquid-liquid microextraction method - A review, Talanta, 190 (2018) 335-356
C. M. Monzón, C. M. Teglia, M. R. Delfino, H. C. Goicoechea, Chemometric optimization and validation of a novel dispersive liquid-liquid microextraction-HPLC method for gliclazide, glibenclamide and glimepiride quantitation in serum samples, Microchem. J., 127 (2016) 113–119.
S. Veyseh, A. Niazi, Talanta A novel aeration-assisted homogenous liquid – liquid microextration for determination of thorium and uranium in water and hair samples by inductively coupled plasma-mass spectroscopy, Talanta, 147 (2016) 117–123.
J. I. Cacho, N. Campillo, P. Viñas, M. Hernández-Córdoba, In situ ionic liquid dispersive liquid-liquid microextraction coupled to gas chromatography-mass spectrometry for the determination of organophosphorus pesticides, J. Chromatogr. A, 1559 (2018) 95–101.
H. Malekzadeh, M. H. Fatemi, Analysis of flavor volatiles of some Iranian rice cultivars by optimized static headspace gas chromatography-mass spectrometry, J. Iran. Chem. Soc., 12 (2015) 2245–2251.
V. Kostik, Development and validation of a method for the simultaneous determination of 20 organophosphorus pesticide residues in corn by accelerated solvent extraction and gas chromatography with nitrogen phosphorus detection, Am. J. Appl. Chem., 2 (2014) 46.
A. Bidari, M. R. Ganjali, P. Norouzi, M. R. M. Hosseini, Y. Assadi, Sample preparation method for the analysis of some organophosphorus pesticides residues in tomato by ultrasound-assisted solvent extraction followed by dispersive liquid-liquid microextraction, Food Chem.,126 (2011)1840–1844.
J. Sherma, Review of thin-layer chromatography in pesticide analysis: 2014–2016, J. Liquid Chromatogr. Related Technol., 40 (2017) 226–238.
J. Hassan, M. Sarkouhi, Miniaturized counter current liquid-liquid extraction for organophosphorus pesticides determination, Arab. J. Chem., 9 (2016) 38–42.
T. T. Hu, C. M. Lu, H. Li, Z. X. Zhang, Y. H. Zhao, J. Li, Determination of Eleven organophosphorus pesticide residues in textiles by using HPLC-HRMS, Anal. Sci., 33 (2017) 1027–1032.
N. Ferretto, M. Tedetti, C. Guigue, S. Mounier, R. Redon, M. Goutx, Identification and quantification of known polycyclic aromatic hydrocarbons and pesticides in complex mixtures using fluorescence excitation-emission matrices and parallel factor analysis, Chemosphere, 107 (2014) 344–353.
J. Dong, P. Li, S. Wang, J. Wei, Y. Yang, Fluorometric determination of pesticides and organophosphates using nanoceria as a phosphatase mimic and an inner filter effect on carbon nanodots, Microchim. Acta, 186 (2019) 66.
H. C. Liang, N. Bilon, M. T. Hay, Analytical methods for pesticide residues, Water Environ. Res., 86 (2014) 2132–2155.
W. M. Marget, M. D. Morris, Central composite experimental designs for multiple responses with different models, Technometrics, 61 (2019) 524-532.
[ 32] N. Khorshidi, A. Niazi, Optimization of pyrocatechol violet biosorption by Robinia pseudoacacia leaf powder using response surface methodology: Kinetic, isotherm and thermodynamic studies, J. Water Reuse Desal., 6 (2016) 333–344.
Z. Zhang, H. Zheng, Optimization for decolorization of azo dye acid green 20 by ultrasound and H2O2 using response surface methodology, J. Hazard. Mater., 172 (2009) 1388–1393.
F. B. Shahri, A. Niazi, Synthesis of modi fi ed maghemite nanoparticles and its application for removal of Acridine Orange from aqueous solutions by using Box-Behnken design, J. Magn. Magn. Mater., 396 (2015) 318–326.
M. H. Dehghani, Production and application of a treated bentonite–chitosan composite for the efficient removal of humic acid from aqueous solution, Chem. Eng. Res. Design, 140 (2018) 102–115.
S. G. Tuncel, T. Topal, Multifactorial optimization approach for determination of polycyclic aromatic hydrocarbons in sea sediments of Turkish Mediterranean Coast, Am. J. Anal. Chem. 2 (2011) 783–794.
R. Bhaumik, N. K. Mondal, S. Chattoraj, J. K. Datta, E. T. Al, Application of Response Surface Methodology for Optimization of Fluoride Removal Mechanism by Newely Developed Biomaterial, Am. J. Anal. Chem., 4 (2013 404–419.
M. Z. Mohamad Zulhelmi, A. Rasyidah, S. Faraziehan, K. Mohamad Anuar, Response surface methodology approach for optimization of biosorption process for removal of binary metals by immobilized saccharomyces cerevisiae, Appl. Mech. Mater., 661(2014) 51–57.
X. Yan, H. Li, X. Wang, X. Su, A novel fluorescence probing strategy for the determination of parathion-methyl, Talanta, 131(2015) 88–94.
M. L. Nahorniak, K. S. Booksh, Optimizing the implementation of the PARAFAC method for near-real time calibration of excitation–emission fluorescence analysis, J. Chemom., 17 (2004) 608–617.
S. Elcoroaristizabal, A. De Juan, J. Antonio, I. Elorduy, N. Durana, L. Alonso, Chemometric determination of PAHs in aerosol samples by fluorescence spectroscopy and second-order data analysis algorithms, J. Chemom., 28 (2014) 260–271.
A. Niazi, M. Sadeghi, PARAFAC and PLS applied to spectrophotometric determination of tetracycline in pharmaceutical formulation and biological fluids, Chem. Pharm. Bull., 54 (2006) 711–713.
J. Ghasemi, A. Niazi, Two- and three-way chemometrics methods applied for spectrophotometric determination of lorazepam in pharmaceutical formulations and biological fluids, Anal. Chim. Acta, 533 (2005)169–177.
K. Kumar, A. Kumar Mishra, Parallel factor (PARAFAC) analysis on total synchronous fluorescence spectroscopy (TSFS) data sets in excitation-emission matrix fluorescence (EEMF) layout: Certain practical aspects, Chemom. Intell. Lab. Syst., 147 (2015) 121–130.
J. U. Ghasemi, A. Niazi, Simultaneous determination of cobalt and nickel, comparison of prediction ability of PCR and PLS using original, first and second derivative spectra, Microchem. J., 68 (2001) 1-11.
A. Niazi, M. Goodarzi, Orthogonal signal correction–partial least squares method for simultaneous spectrophotometric determination of cypermethrin and tetramethrin. Spectrochim. Acta Part A, 62 (2008)1165-1169.
S. Wold, M. Sjöström, L. Eriksson, PLS-regression: A basic tool of chemometrics, Chemom. Intell. Lab. Syst., 58 (2001) 109–130.
Copyright (c) 2021 Analytical Methods in Environmental Chemistry Journal

This work is licensed under a Creative Commons Attribution 4.0 International License.
JOURNAL PUBLISHING AGREEMENT
PLEASE PROVIDE US THE FOLLOWING INFORMATION,
Article entitled:
Corresponding author:
To be published in the journal:
Your Status
I am the sole author of the manuscript
- I am an Iranian government employee.
- I am a European government employee
- I am a Asian government
- None of the above
I am one author signing on behalf of all co-authors of the manuscript
- I am an Iranian government employee.
- I am a European government employee
- I am a Asian government
- None of the above
Please tick the above blanks (as appropriate), review the Journal Publishing Agreement, and then sign and date the document in black ink.
Published Journal Article: the author may share a link to the formal publication through the relevant DOI. Additionally theses and dissertations which contain embedded Published Journal Articles as part of the formal submission may be hosted publicly by the awarding institution with a link to the formal publication through the relevant DOI. Any other sharing of Published Journal Articles is by agreement with the publisher only.
Signed: ______________________________________ Name printed: ___________________________________________
Title and Company (if employer representative): _______________________Date: __________________________________