Mercury determination in work place air and human biological samples based on dispersive liquid-liquid micro-extraction coupled with cold vapor atomic absorption spectrometry
Vol 2, Issue 04, Pages 49-58,*** Field:Air pollution analysis
Abstract
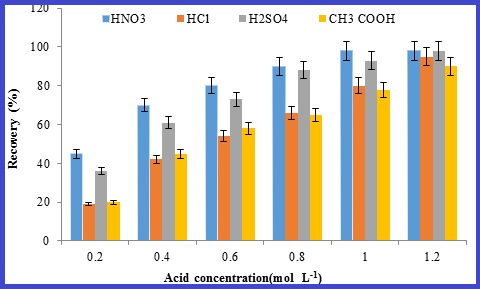
Mercury as a Heavy metal is important factor must be determined and controlled in environmental air and biological. Mercury (Hg) accumulate in living tissues of human body. By NIOSH method, the briefing work place air of petrochemical worker was measured by cold vapor atomic absorption spectrometry(CV-AAS). For preparation and preconcentration samples before analysis a new method of dispersive liquid-liquid micro-extraction with ammonium salt of 1-pyrrolidinedithiocarbamate(APTC)combined with cold vapor atomic absorption spectroscopy, was developed.The influences of various analytical parameters including pH, APDC concentration and ionic liquid volume were investigated.Quantitative recoveries were obtained at pH 7.The enrichment factor was calculated as 5.The detection of limit(LOD)and detection of quantification(LOQ)of mercury were 0.06 and 0.2 μg L-1 respectively. In order accuracy determination the method,certified reference materials, NIST SRM 3133 Lot 061204 was analyzed and the recovery value 98% obtained. So, method of DLLME-APTC had good potential for preparation and preconcentration mercury samples before analysis.
References
WHO. Elemental Mercury and Inorganic Mercury Compounds, Human Health Aspects, Concise International Chemical Assessment Document 50. World Health Organization, Geneva, 2003.
T. Ohno, M. Sakamoto, T. Kurosawa, M. Dakeishi, T. Iwata, K. Murata, Total mercury levels in hair, toenail and urine among women free from occupational exposure and their relations to renal tubular function, Environ. Res., 103 (2007)191-198.
A. Kingman, T. Albertini, LJ. Brown, Mercury concentrations in urine and whole blood associated with amalgam exposure in a US military population. J. Dent. Res., 77 (1998) 461-471.
M.Kampa, E. Castanas, Human health effects of air pollution, In. Environ Pollut., 151 (2008) 362-367.
LL. Booze, Elemental mercury exposure: an evidence-based consensus guideline for out-of-hospital management, Clin. Toxicol., 46 (2008) 21-28.
SC. Foo, Neurobehavioral effects in Occupational Chemical Exposure. Environ. Res., 60 (1993) 267-73.
WHO, Mercury environmental aspects, environmental health criteria. world health organization, Geneva, 2008.
IARC, beryllium, cadmium, mercury and exposures in the glass manufacturing industry, mercury and mercury compounds, International Agency for Research on Cancer, Vol 58, 1997.
H V. Warren, S J. Horksy, C E Gould, Quantitative analysis of zinc, copper, lead, molybdenum, bismuth, mercury and arsenic in brain and other tissues from multiple sclerosis and non-multiple sclerosis cases, Sci. Total Environ., 29 (1983) 163-171.
T W. Clarkson, L. Magos, G J. Myers, The toxicology of mercury current exposures and clinical manifestation, New Eng. J. Med., 349 (2003) 1731-1737.
M E. Vahter, N K Mottet, L T. Friberg, S B. Lind, J S. Charleston, T M. Burbacher, Demethylation of methyl mercury in different brain sites of Macaca fascicularis monkeys during long-term subclinical methyl mercury exposur, Toxicol. Appl. Pharm., 134 (1995) 273-284.
M B. Pedersen, J C. Hansen, G. Mulvad, H S. Pedersen, M Gregersen, G. Danscher, Mercury accumulations in brains from populations exposed to high and low dietary levels of methyl mercury. Concentration, chemical form and distribution of mercury in brain samples. Int. J. Circumpolar Health, 58 (1999) 96-107.
M. Nylander, L. Friberg, J Weiner, Muscle biopsy as an indicator for predicting mercury concentrations in the brain, Br. J. Ind. Med., 47 (1990) 575-576.
G. Turabelidze, Multiple sclerosis prevalence and possible lead exposure. J. Neurol. Sci., 269 (2009) 158-162.
OSHA, Guidelines for mercury vapor. Occupational safety and health administration, 2008. Available at: www.osha-slc.gov
NIOSH, Centers for disease control and prevention. National Institute for Occupational Safety and Health, guide to chemical hazards, 3th Ed, 2007.
ACGIH, U.S Documentation of the threshold limit values and biological exposure. American Conference of Governmental Industrial Hygienists, 7th Ed, 2011.
N M. Tietz, Clinical Guide to Laboratory Tests. W. B. Saunders, Philadelphia, PA, 8rd Ed, 2010.
NIOSH, Documentation of the threshold limit values and biological exposure indices. National Institute for Occupational Safety and Health, Cincinnati, OH., 7th Ed., 2001.
Y. Cai, Speciation and analysis of mercury, arsenic, and selenium by atomic fluorescence spectrometry, TrAC Trends Anal. 19 (2000) 62-66.
Gao, Ying, Determination and speciation of mercury in environmental and biological samples by analytical atomic spectrometry, Microchem. J., 103 (2012) 1-14.
J. Szkoda, J. Zmudzki, A. Grzebalska, "Determination of total mercury in biological material by atomic absorption spectrometry method, J. Bull. Vet. Inst. Pulawy, 50 (2006) 363.
H. Erxleben, R. Jaromir, Atomic absorption spectroscopy for mercury, automated by sequential injection and miniaturized in lab-on-valve system, Anal. Chem., 77.16 (2005) 5124-5128.
W. Peng, H. Liang, Z Chengbin, H. Xiandeng, E S Ralph, Applications of chemical vapor generation in non-tetrahydroborate media to analytical atomic spectrometry, J. Anal. Atom. Spec., 25.8 (2010) 1217-1246.
CF. Harrington, the speciation of mercury and organomercury compounds by using high-performance liquid chromatography, TrAC, Trends Anal. Chem., 19 (2000) 167-179.
CJ, Cappon, J C Smith, Gas-chromatographic determination of inorganic mercury and organomercurials in biological materials, Anal. Chem., 49.3 (1977) 365-369.
SCK. Shum, HM. Pang, RS Houk, Speciation of mercury and lead compounds by microbore column liquid chromatography-inductively coupled plasma mass spectrometry with direct injection nebulization, Anal. Chem., 64 (1992) 2444-2450.
L. N. Liang, G. B. Jiang, J. F. Liu, J. T. Hu, Speciation analysis of mercury in seafood by using high-performance liquid chromatography on-line coupled with cold-vapor atomic fluorescence spectrometry via a post column microwave digestion, Anal. Chim. Acta, 477 (2003) 131-137.
X Jia, Y Han, X Liu, T Duan, H Chen, Speciation of mercury in water samples by dispersive liquid–liquid microextraction combined with high performance liquid chromatography-inductively coupled plasma mass spectrometry, Spectrochim. Acta Part B: Atom. Spec., 66 (2011) 88-92.
S. Mishra, R. M. Tripathi, S. Bhalke, V. K. Shukla, V. D. Puranik, Determination of methylmercury and mercury (II) in a marine ecosystem using solid-phase microextraction gas chromatography-mass spectrometry, Anal. Chim. Acta, 551 (2005) 192-198.
Q. Liu, Determination of mercury and methylmercury in seafood by ion chromatography using photo-induced chemical vapor generation atomic fluorescence spectrometric detection, Microchem. J., 95 (2010) 255-258.
M. Park, H. Yoon, C. Yoon, J. Y. Yu, Estimation of mercury speciation in soil standard reference materials with different extraction methods by ion chromatography coupled with ICP-MS, Environ. Geochem. health, 33 (2011) 49-56.
S. Carneado, R. Peró-Gascón, C. Ibáñez-Palomino, J. F. López-Sáncheza, A. Sahuquillo, Mercury (II) and methylmercury determination in water by liquid chromatography hyphenated to cold vapour atomic fluorescence spectrometry after online short-column preconcentration, Anal. Method., 7 (2015) 2699-2706.
A. Fashi, MR. Yaftian, A. Zamani, Electromembrane extraction-preconcentration followed by microvolume UV–Vis spectrophotometric determination of mercury in water and fish samples, Food chem., 221 (2017) 714-720.
D. M, Sánchez, R. Martı́n, R. Morante, J. Marı́n, M.L. MunueraPreconcentration speciation method for mercury compounds in water samples using solid phase extraction followed by reversed phase high performance liquid chromatography, Talanta, 52 (2000) 671-679.
M. Krawczyk, E. Stanisz, Ultrasound-assisted dispersive micro solid-phase extraction with nano-TiO2 as adsorbent for the determination of mercury species, Talanta, 161 (2016): 384-391.
E. Ziaei, A. Mehdinia, A. Jabbari, A novel hierarchical nanobiocomposite of graphene oxide–magnetic chitosan grafted with mercapto as a solid phase extraction sorbent for the determination of mercury ions in environmental water samples, Anal. Chim. Acta, 850 (2014) 49-56.
N. Pourreza, K. Ghanemi, Determination of mercury in water and fish samples by cold vapor atomic absorption spectrometry after solid phase extraction on agar modified with 2-mercaptobenzimidazole, J. Hazard. Mater., 161 (2009) 982-987.
A Krata, K Pyrzyńska, E Bulska, Use of solid-phase extraction to eliminate interferences in the determination of mercury by flow-injection CV AAS, Anal. Bioanal. Chem., 377 (2003) 735-739.
L. Adlnasab, H. Ebrahimzadeh, A. A. Asgharinezhad, M. Nasiri Aghdam, A. Dehghani, S. Esmaeilpour, preconcentration procedure for determination of ultra-trace mercury (II) in environmental samples employing continuous-flow cold vapor atomic absorption spectrometry, Food anal. Method., 7 (2014) 616-628.
NH Khdary, AG Howard, New solid-phase-nanoscavenger for the analytical enrichment of mercury from water, Analyst, 136 (2011) 3004-3009.
E. Zolfonoun, A. Rouhollahi, A. Semnani, Solid-phase extraction and determination of ultra-trace amounts of lead, mercury and cadmium in water samples using octadecyl silica membrane disks modified with 5, 5′-dithiobis (2-nitrobenzoic acid) and atomic absorption spectrometry, Int. J. Environ. Anal. Chem., 88 (2008) 327-336.
Y. Date, et al, Trace-level mercury ion (Hg2+) analysis in aqueous sample based on solid-phase extraction followed by microfluidic immunoassay, Anal. Chem., 85 (2012) 434-440.
P. Rivaro, C. Ianni, F. Soggia, R. Frache, Mercury speciation in environmental samples by cold vapour atomic absorption spectrometry with in situ preconcentration on a gold trap, Microchim. Acta, 158 (2007) 345-352.
H. Shirkhanloo, A. Khaligh, H. Zavvar Mousavi, M. M. Eskandari, A. A. Miran-Beigi, Ultra-trace arsenic and mercury speciation and determination in blood samples by ionic liquid-based dispersive liquid-liquid microextraction combined with flow injection-hydride generation/cold vapor atomic absorption spectroscopy, Chem. Paper.. 69 (2015) 779-790.
N. Rajesh, G. Gurulakshmanan, Solid phase extraction and spectrophotometric determination of mercury by adsorption of its diphenylthiocarbazone complex on an alumina column, Spectrochim. Acta Part A: Mol. Biomol. Spec., 69.2 (2008) 391-395.
HI. Ulusoy, R Gürkan, S Ulusoy, Cloud point extraction and spectrophotometric determination of mercury species at trace levels in environmental samples, Talanta, 88 (2012) 516-523.
J. C. de Wuilloud, R. G. Wuilloud, M. F. Silva, R.A. Olsina, L. D. Martinez, Sensitive determination of mercury in tap water by cloud point extraction pre-concentration and flow injection-cold vapor-inductively coupled plasma optical emission spectrometry, Spectrochim. Acta Part B: Atom. Spec., 57 (2002) 365-374.
A. Nasrollahpour, S. M. J. Moradi, S. E. Moradi. Dispersive solid phase micro-extraction of mercury (II) from environmental water and vegetable samples with ionic liquid modified graphene oxide nanoparticles, J. Serb. Chem. Soc., 82 (2017): 551-565.
C. Mitani, A. Kotzamanidou, A.N. Anthemidis, Automated headspace single-drop microextraction via a lab-in-syringe platform for mercury electrothermal atomic absorption spectrometric determination after in situ vapor generation, J. Anal. Atom. Spec., 29 (2014) 1491-1498.
M. Hossien-poor-Zaryabi, et al. "Application of dispersive liquid–liquid micro-extraction using mean centering of ratio spectra method for trace determination of mercury in food and environmental samples. Food Anal. Method., 7 (2014): 352-359.
M. Rezaee, Y. Assadi, M. R. M. Hosseini, E. Aghaee, F. Ahmadi, S. Berijani, Determination of organic compounds in water using dispersive liquid–liquid microextraction, J. Chromatogr. A, 1116 (2006) 1-9.
G. Li, K. H. Row, Utilization of deep eutectic solvents in dispersive liquid-liquid micro-extraction, TrAC, Trends Anal. Chem., 120 (2019) 115651.
Li, Yanyan, et al, Magnetic effervescent tablet-assisted ionic liquid-based dispersive liquid-liquid microextraction of polybrominated diphenyl ethers in liquid matrix samples, Talanta, 195 (2019) 785-795.
Copyright (c) 2019 Analytical Methods in Environmental Chemistry Journal

This work is licensed under a Creative Commons Attribution 4.0 International License.
JOURNAL PUBLISHING AGREEMENT
PLEASE PROVIDE US THE FOLLOWING INFORMATION,
Article entitled:
Corresponding author:
To be published in the journal:
Your Status
I am the sole author of the manuscript
- I am an Iranian government employee.
- I am a European government employee
- I am a Asian government
- None of the above
I am one author signing on behalf of all co-authors of the manuscript
- I am an Iranian government employee.
- I am a European government employee
- I am a Asian government
- None of the above
Please tick the above blanks (as appropriate), review the Journal Publishing Agreement, and then sign and date the document in black ink.
Published Journal Article: the author may share a link to the formal publication through the relevant DOI. Additionally theses and dissertations which contain embedded Published Journal Articles as part of the formal submission may be hosted publicly by the awarding institution with a link to the formal publication through the relevant DOI. Any other sharing of Published Journal Articles is by agreement with the publisher only.
Signed: ______________________________________ Name printed: ___________________________________________
Title and Company (if employer representative): _______________________Date: __________________________________